BIOELECTRIC CUSTOMIZED
INFLAMMATION CONTROL
world’s first designed to read
inflammation real time and constantly adjust
bioelectric stimulation and cytokine
releases to modulate and manage
inflammation real time.
InStim – Patent Pending Technology
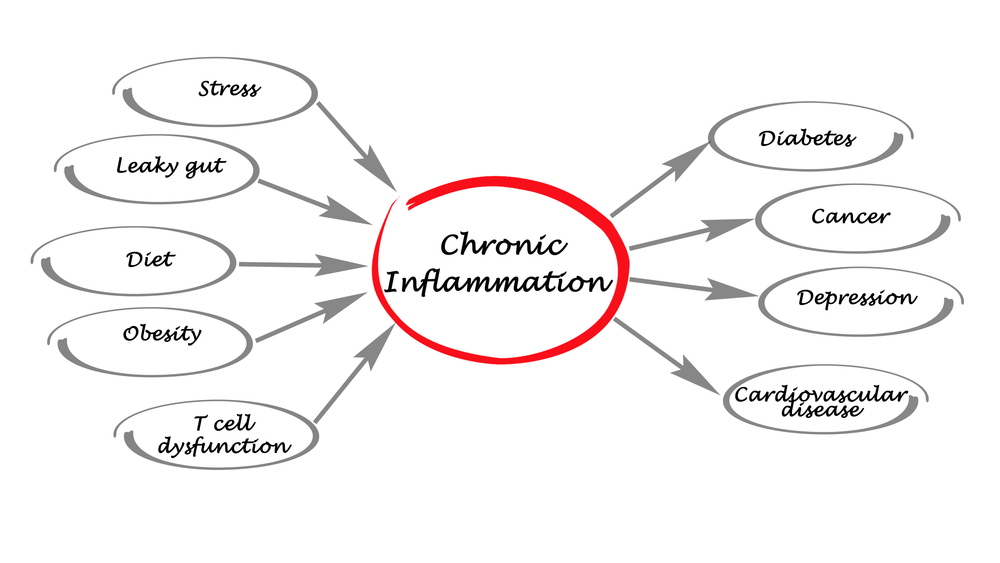
InStim’s patent pending technology is the world’s first designed to read inflammation real time and constantly adjust bioelectric stimulation and cytokine releases to modulate and manage inflammation real time. We believe this is the only way to properly break the cycle of chronic inflammation.
We believe we can hit a re-start button on a person’s chronic inflammation cycle!
Our microprocessor stores ideal optimal healing cycles and the treatment regimes are adjusted real time between data coming in via real-time monitoring of inflammation and ideal optimal healing algorithms stored in our microprocessor of our micro stimulator + reader. The ideal optimal healing cycles are derived from measurements made in healthy subjects that have an inflammation-causing event and recover quickly and fully (such as a toddler head injury recovery).
Get in touch.
